G. Mastronardi, J. Perego, C. X. Bezuidenhout, W. Temmerman, V. Van Speybroeck, V. Crocellà, S. Bracco, N. Li Pira, A. Comotti and S. Bordiga
Small 2026, 22:e14197
Abstract
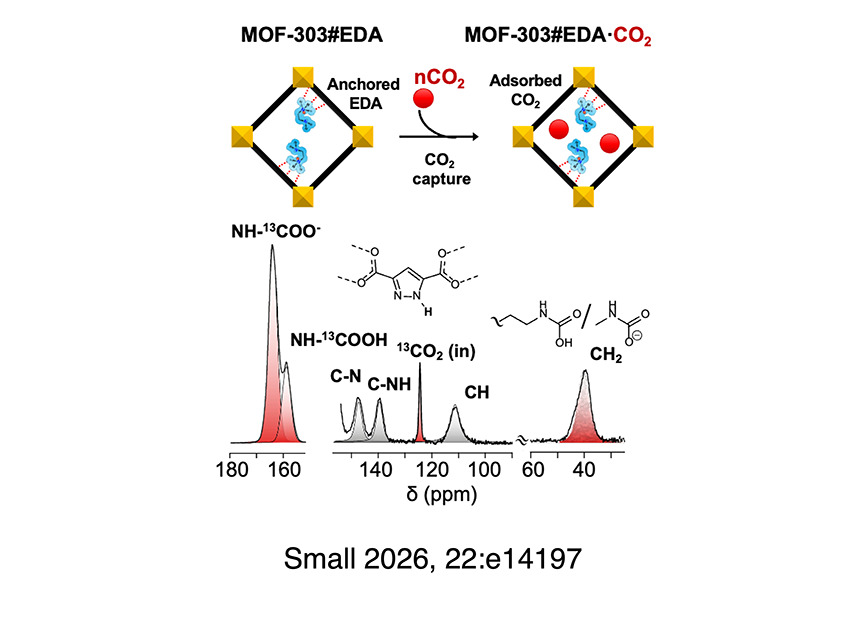
Amine-functionalized materials have been extensively investigated for CO2 capture, with adsorption mechanisms based primarily on carbamate formation. Among the different strategies, grafting amine moieties onto porous solid supports represents a benchmark approach for developing highly effective adsorbents, even at very low CO2 concentrations. In this work, we propose a novel strategy for anchoring amine functionalities onto the surface of a Metal–Organic Framework (MOF), exploiting the unique chemistry/reactivity of the inexpensive and scalable MOF-303. The pyrazole linkers in MOF-303 serve as acidic anchoring sites for ethylenediamine molecules (EDAs), protonating one of the two amines to form an ammonium cation, which is then grafted to the pyrazolate moieties through charged-assisted hydrogen bonds. The resulting MOF-303#EDA exhibits a remarkable CO2 uptake of 0.71 mmol g−1 at 298 K and 450 ppm CO2. Notably, the high CO2 uptake of this material at 0.15 bar (2.5 mmol g−1) and its cyclability demonstrated in breakthrough experiments make it a promising candidate for point source CO2 capture industrial applications. The complex nature of amine grafting on MOF-303 and its interaction with CO2 are investigated in depth by combining spectroscopic techniques (FT-IR and SS-NMR), synchrotron-source X-ray diffraction, and computational modeling.