A. Rigamonti, C. X. Bezuidenhout, J. Perego, E. Montanari, A. Comotti and S. Bracco
Chem. Mater. 2026
Abstract
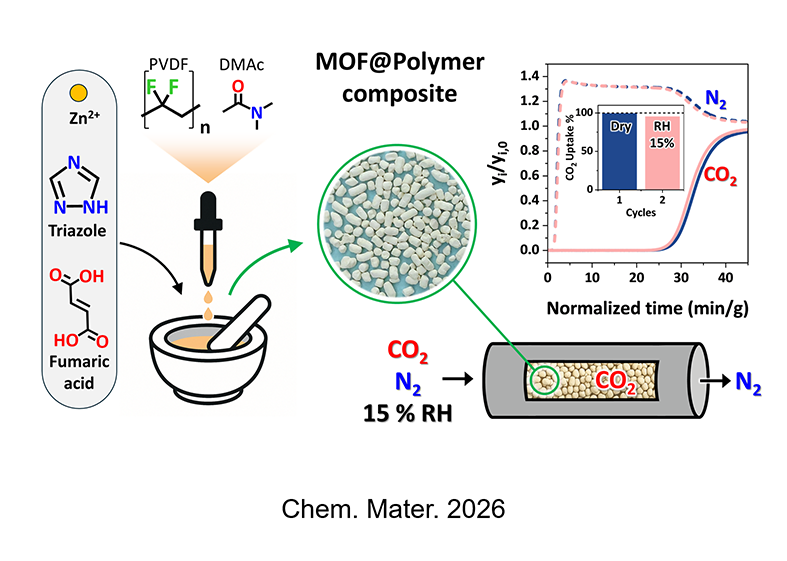
Pillared-layer metal–organic frameworks (MOFs) comprising triazolate and oxalate or organodicarboxylate linkers are emerging materials for selective CO2 capture in large-scale applications. One key aspect to consider for industrial applications is the search for synthetic strategies that minimize the use of solvents, thereby reducing the environmental impact and improving scalability. Herein, a protocol that combines liquid-assisted mechanochemistry and incubation with a minimal amount of solvent was applied to produce both a triazolate-fumarate Zn-MOF and a MOF-polymer composite shaped into self-supporting cylindrical objects amenable to CO2 capture even under humid conditions. The Zn-MOF exhibits the highest surface area and CO2 adsorption capacity in this class of materials, and a CO2/N2 selectivity of 200 at 273 K. A 36 kJ/mol value of heat of adsorption was estimated by direct measurement of the heat exchanged upon stepwise CO2 loading, in tandem with a CO2 adsorption isotherm. Upon one hundred CO2 adsorption/desorption cycles, the Zn-MOF exhibits remarkable cycling stability. Breakthrough experiments with CO2/N2 mixtures containing 5, 10, and 15% CO2 unveiled selective CO2 adsorption even in the presence of humidity (up to 15%) and full recovery of the composite sorption performance through solvent incubation. In-depth spectroscopic and diffractometric characterization of the activated material and its composite revealed the sophisticated and dynamic arrangement of pillars and layers within the framework. In situ powder X-ray diffraction (PXRD) during CO2 loading and unloading, combined with ab initio computational studies, enabled the determination of the preferred CO2 adsorption sites within the framework.